FDA approves first radiology diagnostics app
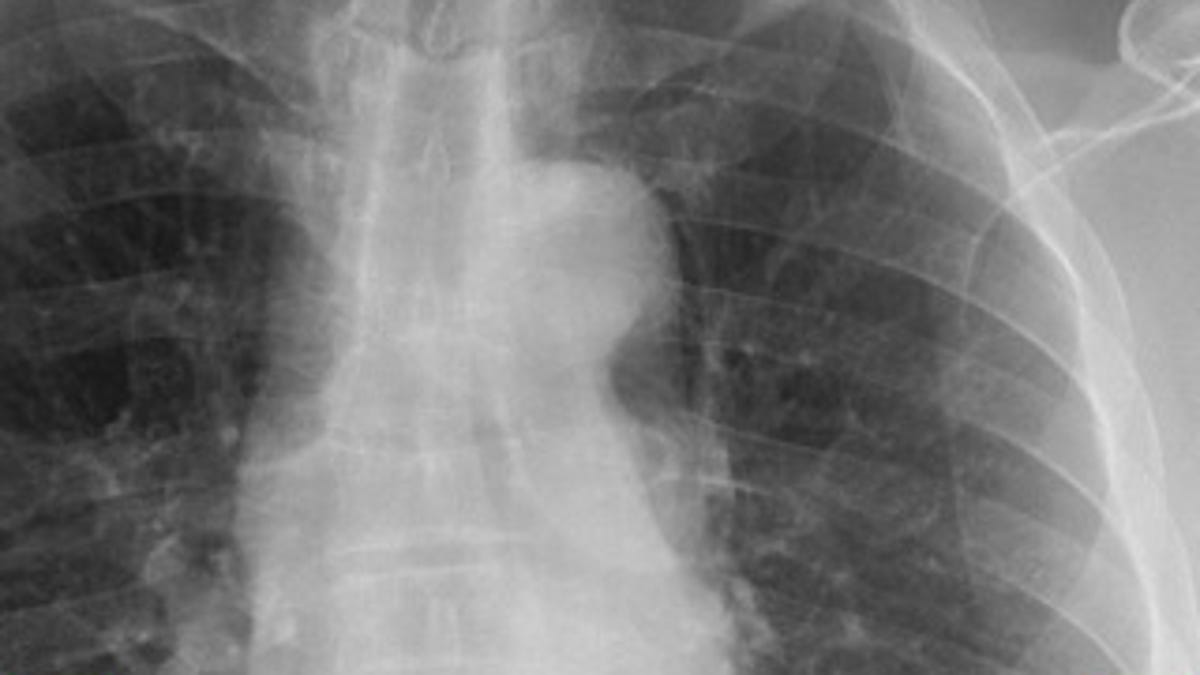
Mobile MIM lets physicians use an iPad or iPhone to measure distances and image intensity in CT, PET, MRI, and other scans, as well as display measurement lines and notes.
There is no shortage of health-related apps. Some 1,500 cater to professional caregivers and laypersons alike for a range of purposes, from counting calories to learning anatomy or pulling up drug dosage recommendations.
But Mobile MIM is the first mobile app to be cleared (just last week) by the U.S. Food and Drug Administration that allows physicians to make medical diagnoses using images transmitted to their iPhones or iPads.
The app transmits several image types, including those from computed tomography (CT), magnetic resonance imaging (MRI), and nuclear medicine technology such as positron emission tomography (PET). Using software developed by Cleveland-based MIM Software, Mobile MIM allows for not just viewing of medical images, but also displaying measurement lines, annotations, and areas of interest.
While the app is indicated for use only in the absence of a workstation, it's pretty clear that the ability to view radiology images on the go could result in, shall we say, dusty workstations.
"This important mobile technology provides physicians with the ability to immediately view images and make diagnoses without having to be back at the workstation or wait for film," announced William Maisel, chief scientist and deputy director for science in the FDA's Center for Devices and Radiological Health, in a statement.
The app includes a screen contrast test to address a major concern of display performance in mobile devices--that variations in luminance levels even within the same model, not to mention environmental lighting, could result in insufficient quality for diagnosing. To test quality, each image includes a small section that is a slightly different shade than the rest; identify and tap on that section and image quality is confirmed as sufficient for discerning subtle contrast differences.
We've been waiting for Mobile MIM since it won the Apple Design Award for Best iPhone Healthcare & Fitness Application in 2008. It's already available in 14 languages via the Apple app store if purchased from any of 34 countries throughout Europe, Asia, Latin America, and the Middle East. It is expected to be available for U.S. consumers sometime this week. Pricing details should be released shortly.

