Coronavirus antibody, nasal swab, saliva testing: What to know about COVID-19 tests
Coronavirus testing will be an important tool for lifting restrictions. Here's a look at the COVID-19 tests available now or in the works.
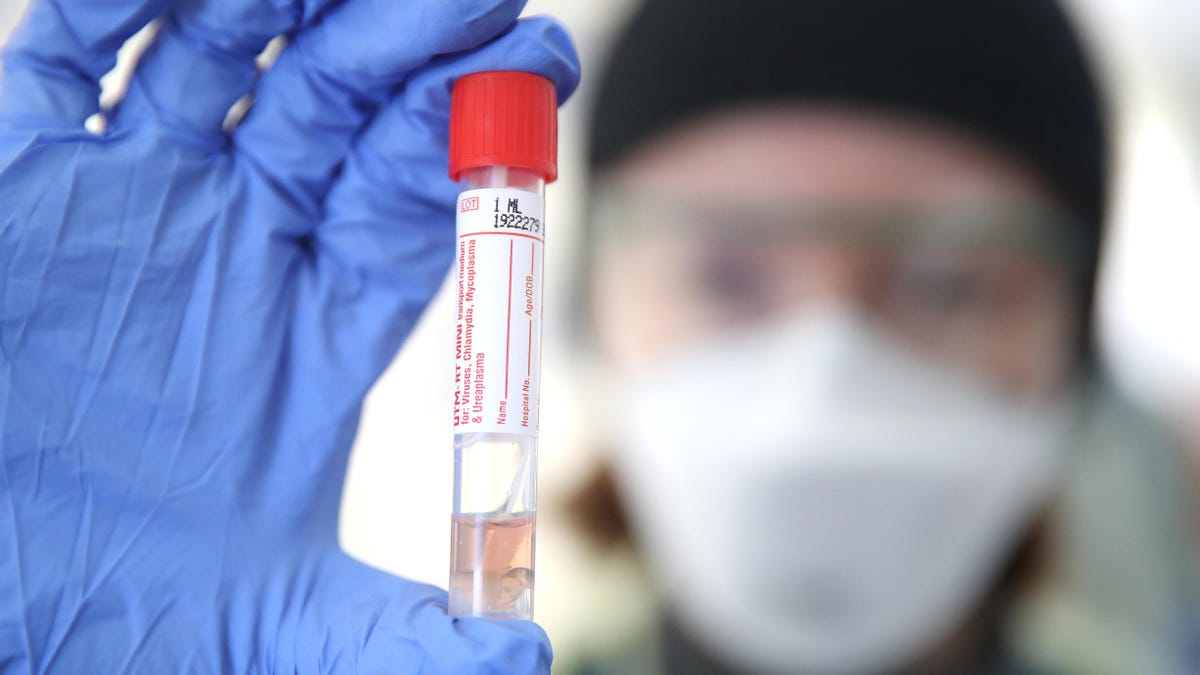
How will you get tested for COVID-19?
Coronavirus testing broadly available could be the key to returning to life as usual. That's the thought, anyway, as health authorities from cities to countries rush to test as many people as they can. Testing can help determine who has already been infected and therefore might be less likely to contract or spread the coronavirus again. Understanding who tests positive for the COVID-19 disease can also help scientists better understand disease patterns and help officials monitor new outbreaks.
There are several types of tests available now and some being worked on for the future: the nose swab, antibody testing, a saliva test and a promising method that uses DNA to diagnose you in about 40 minutes, rather than hours.
Right now, because test kits are still so limited, testing for COVID-19 requires a doctor's referral in many places, and prioritizes patients who are sickest. As labs ramp up their production of Food and Drug Administration-approved coronavirus tests kits, community-wide testing will get underway.
We'll tell you what you need to know about the different types of coronavirus tests, based on guidance from the FDA and Centers for Disease Control and Prevention. Access to tests will vary by regions, with some testing sites already coming online. Apple Maps can help you find testing locations. Here's how to know if you qualify for a test and why there aren't any coronavirus at-home test kits yet.
We'll update this story frequently as test kits become more widely available over the next few months. For now, here is a snapshot of the types of testing available.
Coronavirus updates
Nose swab test
What it is: Right now, this is the most common test for COVID-19, and is recommended by the CDC. In the nasal (or nasopharyngeal) swab test, a doctor will stick a long Q-tip-like swab into one or both nostrils to collect a sample. The process is described as being slightly uncomfortable but not painful. Related is the throat, or oropharyngeal, test swab, which takes a sample from the back of the throat.
How it's used now: The test is being used mainly to detect the coronavirus in high-risk situations, such as with health care professionals or based on an individual's symptoms, age and health, or recent travel history. Once a sample is collected, it can take four hours to run the test. But it may be days or weeks to receive the results because of the time it takes to get the sample to the lab, run the test and then report the results, and labs in some regions are backlogged with testing samples.
A quicker testing nasal-swab method using a tool named CRISPR can produce results in 40 minutes. Another swab test from Abbott Labs can return results in 5 minutes, Abbott says.
Accuracy: The tests are just rolling out, and the FDA has not released numbers for how accurate the test is.
Fighting coronavirus: COVID-19 tests, vaccine research, masks, ventilators and more
See all photosAntibody test
What it is: Coronavirus tests break into two broad categories: Those tests that check for the virus to see if you're currently infected, and those that look for antibodies to see if you've previously been infected and have built up an immune response. Within each of those two broad categories are multiple types of tests that medical labs are working on and rolling out.
An antibody test -- also called a serology test -- can tell if you've previously been infected with COVID-19 and have built up an immune response through antibodies. The test requires a blood sample.
How it's used now: Until recently, antibody tests were not widely available in the US. Once they are, health care officials hope to use test results to determine who has already been infected and can return work, for example. On a larger scale, test results across the community can reveal how close a region is to reaching herd immunity by previous exposure. Since a coronavirus vaccine is thought to be more than a year away, antibody testing is considered by many to be the path to safely reopening countries.
Accuracy: As with the swab tests, the FDA and testing labs are not yet talking about the accuracy of antibody tests.
Saliva test
What is it: Having just received FDA emergency use authorization, the saliva test developed in collaboration with Rutgers University would allow health care workers to collect samples by having patients spit into a vial -- much like when providing a sample for DNA tests -- instead of undergo a nasal swab.
How it's used now: While the test currently requires a medical professional to take the sample, Rutgers says the test is designed to let people provide a sample at home by spitting into a vial, once the process receives FDA approval for self-collection.
Accuracy: There's not enough information to determine accuracy yet, but researchers think saliva tests could be as accurate as swab tests.
Testing centers can take place in hospitals, converted facilities, even tents.
Take-home test
What is it: The FDA has just authorized the first home test kit, which should be available in the coming weeks. With a take-home test, you receive a kit with doctor approval that you use to take a sample with a swab and then mail it to a lab for testing.
How's it used: The first home kit from LabCorp uses a swab to collect a nasal sample.
Accuracy: FDA Commissioner Dr. Stephen Hahn said he expects results from the first home test kit to be as accurate as those collected at a testing site.
Testing will play a key role in helping authorities decide when to lift restrictions, and here are what steps you need to take to avoid spreading the virus further as society opens back up. If you're still under restrictions, here's what to do to stay safe when you need to go out in public.

