Damaged Liver Is Repaired in Machine for Three Days, Then Transplanted
The liver recipient was a cancer patient running out of time on the transplant waitlist. One year post-procedure, he's still doing well.
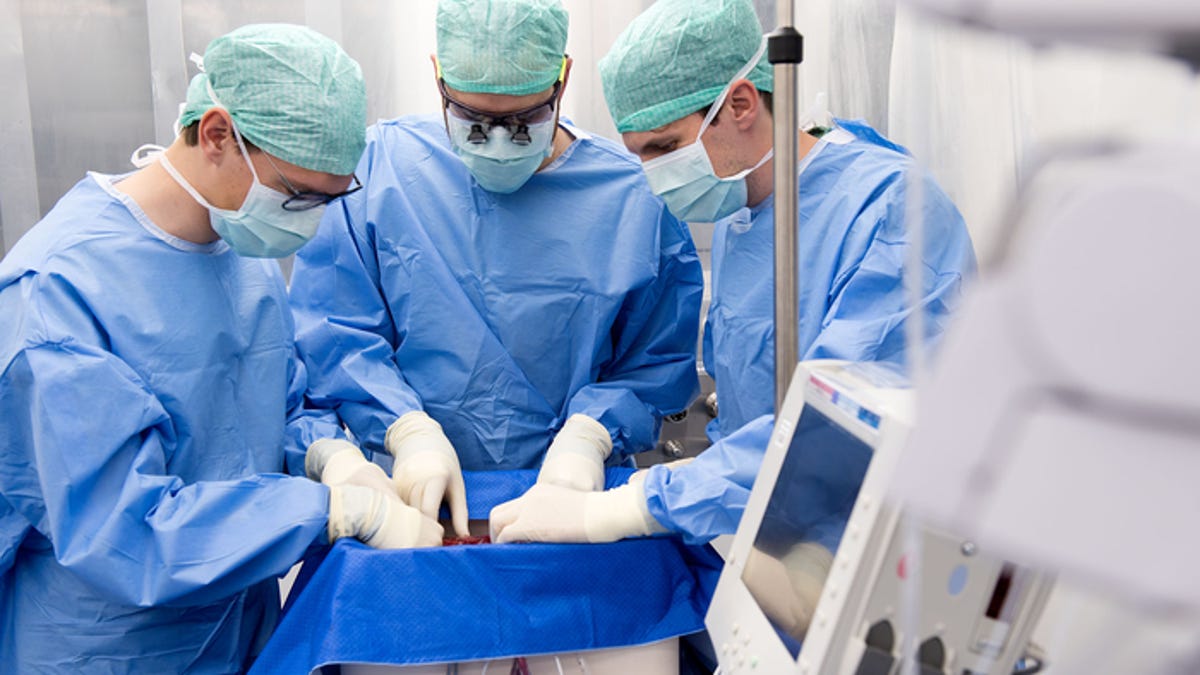
The team connects the donor liver to the perfusion machine in the clean room.
Back in May 2021, Swiss researchers removed the damaged liver of an organ donor, placed it in a novel machine that mimicked the human body's biology and treated it in that machine for three days. They then transplanted the newly repaired organ into a cancer patient who needed a new liver but was stuck on the painfully long organ transplant waitlist.
As of Tuesday, one year since the trailblazing procedure, the team reports that the organ recipient is still doing exceptionally well.
"The patient rapidly recovered a normal quality of life without any signs of liver damage," the research team, called Liver4Life, wrote in a paper about the medical milestone published Tuesday in the journal Nature Biotechnology.
"I am very grateful for the life-saving organ," the 62-year-old organ recipient said in a statement. "Due to my rapidly progressing tumor, I had little chance of getting a liver from the waiting list within a reasonable time." The patient's name was not released.
Dr. Pierre-Alain Clavien, who was the surgeon, stands with the patient (right) as he was discharged from hospital after the successful transplantation last year.
Typically, even perfectly viable donor livers can only remain outside the human body for about 12 hours in conventional cold storage or a maximum of 24 hours in a machine like the team's new one, dubbed a perfusion machine.
Within that time frame, doctors need to "assess, transport and implant donor grafts for human transplantation," per the paper -- not only does this greatly limit the distance these organs can travel before reaching a recipient and room for error during transport, but it also essentially rules out the possibility of repairing an organ prior to transplantation.
Damaged donor organs are often discarded, further exacerbating the organ shortage crisis. According to Organdonor.gov, well over 100,000 Americans are awaiting an organ transplant, and 17 patients die each day while still on the waitlist.
But because the Swiss researchers' new style of perfusion machine sort of acted as a second human body, it allowed the liver specimen to survive externally for three days. They first announced the concept in 2020, at the time stating that their invention can keep a liver outside the human body for one week. However, this marks the first time a liver kept in the device was successfully transplanted into a patient.
"Our therapy shows that treating livers in the perfusion machine makes it possible to alleviate the lack of functioning donor organs and save lives," Dr. Pierre-Alain Clavien, director of the Clinic for Visceral and Transplantation Surgery at the University Hospital Zurich and the surgeon who transplanted the organ, said in a statement.
Basically, this machine has a pump to serve in place of a heart, an oxygenator in place of lungs and dialysis unit in place of kidneys. It can infuse the organs with nutrients and hormones that would normally come from the intestine and pancreas, and even moves the liver to the rhythm of human breathing like a diaphragm would. Additionally, it offers a similar pressure and temperature to natural human biology. In other words, for a liver in the contraption, it's almost like it never left the body.
The perfusion machine replaces the functions of various organs in order to keep the donor liver alive outside of the body.
So in this highly regulated environment, during those three days, the team flushed the liver with antibiotics and hormonal therapies, for instance, while monitoring how well the organ functioned, like in terms of bile production and tissue strength. This went on until the liver met the standards of a "good" donor organ. Then, it was successfully transplanted into the organ recipient with his consent.
"The transplanted liver exhibited normal function, with minimal reperfusion injury and the need for only a minimal immunosuppressive regimen," the paper states. Immunosuppressive medication is vital after transplant procedures because these drugs essentially tell the body's immune system to not attack while the foreign organ gets acquainted with its new world. Without the "don't attack" warning, the body can reject the organ, deeming it an intruder.
Dr. Pierre-Alain Clavien and Dr. Philipp Dutkowski during the transplantation of the liver treated in the machine.
And beyond remarkably saving a life, the team's achievement marks a tremendous breakthrough for the field of medicine. It's direct evidence that damaged donor organs can be repaired and considered for patients awaiting transplants -- transplants that could mean the difference between life and death. Plus, it's proof that one day, transplantation doesn't necessarily have to be an emergency, time crunch procedure.
"This inaugural clinical success opens new horizons in clinical research and promises an extended time window of up to 10 days for assessment of viability of donor organs as well as converting an urgent and highly demanding surgery into an elective procedure," the paper states. Going forth, the team plans to test the procedure on other patients as well as develop the next generation of these powerful perfusion machines.
"The interdisciplinary approach to solving complex biomedical challenges that is used in this project is the future of medicine," Mark Tibbitt, a professor of Macromolecular Engineering at ETH Zurich, said in a statement.

