Philips recalls sleep apnea machines, ventilators
The company cites risks associated with the foam in some devices.
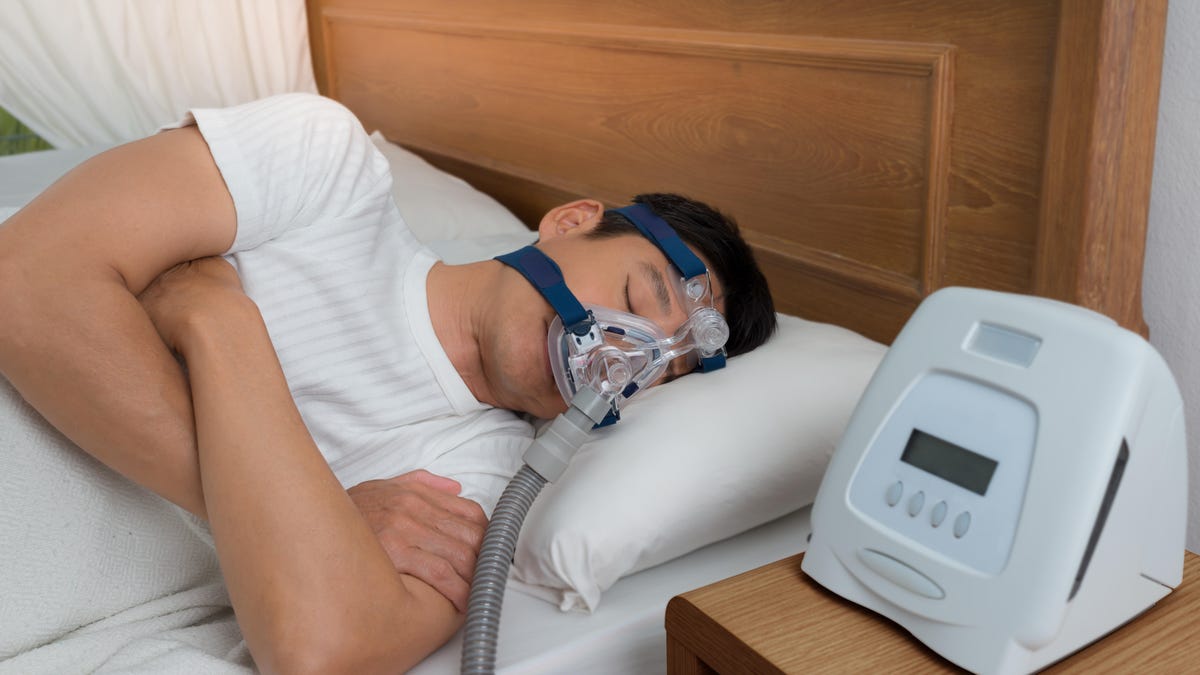
Philips issued a recall earlier this month for several breathing machines and ventilators, citing health risks associated with the foam in certain devices. The affected devices include the Bi-Level PAP, CPAP and mechanical ventilator devices, which are used for sleep apnea and other breathing conditions. Most are DreamStation products, the company said.
The "sound abatement" polyurethane foam used to absorb noise in these devices has been found to pose a risk to users because of the way the foam can "degrade" into particles, which can then be ingested or inhaled, the company said in a press release. Another risk, Philips said, is that the foam may give off gas from certain chemicals.
The exposure to foam particles could cause headache, irritation, inflammation, respiratory issues, and possible toxic and carcinogenic effects, Philips said. Chemical exposure due to off-gassing can cause many of the same symptoms, the company said, along with hypersensitivity, nausea and vomiting.
Philips hasn't reported any deaths associated with its recalled devices, but the company said those with a Bi-Level PAP or CPAP device should discontinue use and consult with a health care provider to determine the most appropriate options for continued treatment. Those who are using a life-sustaining affected device shouldn't stop prescribed therapy, Philips said, until consulting a physician.
The Philips' recall of sleep apnea machines and respiratory care devices is considered a voluntary recall, as are most recalls of medical devices, according to the Food and Drug Administration. Per the agency, a recall is "a method of removing or correcting products that are in violation of laws administered by the Food and Drug Administration."
Philips said it will replace the current foam with a new material and obtain the relevant regulatory clearances.
The company has posted a list of devices covered by the recall. Customers can make a claim by registering their affected device with the company.

