FDA approves single-lead implantable cardiac defibrillator
The device comes with a cellular-based home monitoring system that allows physicians to detect a range of heart-related events, including silent arrhythmias.
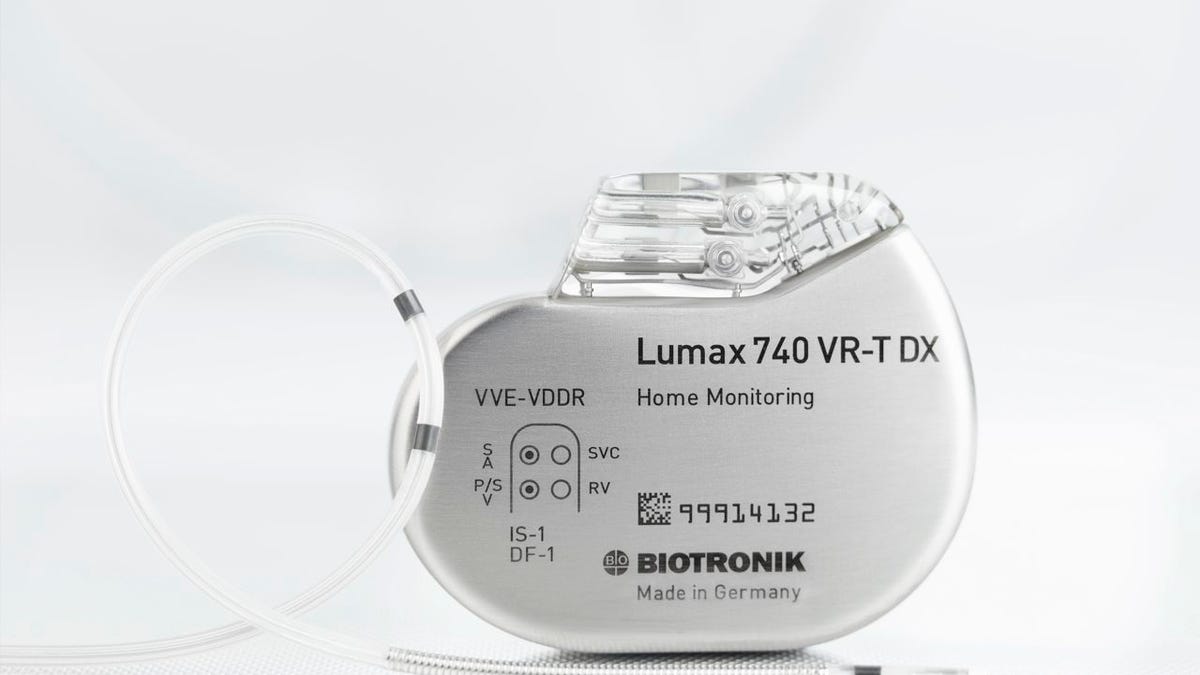
Cardio med tech company Biotronik today announced Food and Drug Administration approval of the world's first implantable cardiac defibrillator that uses just one lead to sense atrial arrhythmias.
Say what?
For those unfamiliar with the mechanics of the heart, let's back up. Atrial fibrillation (AF), the most common heat arrhythmia, occurs when the electrical signals in the atria (the heart's two upper chambers) fire fast and frenetically, causing the atria to essentially quiver instead of pulse regularly, which can result in blood pooling or clotting and thus greatly increase the risk of stroke and congestive heart failure.
As if that's not bad enough, this type of arrhythmia -- which can last for minutes, days, or even be a permanent condition -- is often asymptomatic. This explains why even the most elite young athletes in the world can suddenly drop to the ground mid-performance and suffer a heart attack without ever knowing they were at risk in the first place.
Until now, the only way to constantly monitor patients with or at a heightened risk of AF was to implant a cardiac defibrillator (ICD) that uses two leads (thin, flexible wires) and has pacemaker functions built in to deliver electrical shocks to the heart when it starts beating dangerously fast.
It doesn't take an expert to work out that the risk of complications increases with the amount of hardware implanted in the heart. So patients who don't appear to be at specific risk of AF are typically given a single-lead ICD, which until now hasn't been able to detect AF.
Enter Biotronik's newly approved Lumax 740 DX System, which uses a newfangled lead (called Linoxsmart S DX) that features a floating atrial dipole, along with some fancy algorithms, to differentiate between actual AF and atrial flutter, as well as another type of arrhythmia called supraventricular tachycardias.
In other words, the folks at Biotronik have managed to incorporate atrial sensing into a single-lead ICD.
"Until now, our only option to obtain important and useful atrial signal information from patients undergoing defibrillator implantation has been to implant a separate atrial lead," says Bradley P. Knight, medical director of the Center for Heart Rhythm Disorders at Northwestern Memorial Hospital's Bluhm Cardiovascular Institute. "The DX System addresses a significant gap in ICD therapy. Patients now have access to the benefits of both dual and single-chamber ICDs without the risk of additional hardware."
Biotronik -- which is headquartered in Germany and happens to be celebrating its 50-year anniversary in 2013 -- says it expects the implants will be available in the U.S. in the next few weeks, and that the DX System is currently approved and available in many international markets as well, including Japan and the European Union.
The DX System comes with a cellular-based home monitoring system that enables physicians to monitor the device remotely so that a wide range of important but hard-to-detect events, including silent arrhythmias, can be caught as early as possible for medical intervention.

